Timeline to FSMA 204 Compliance, Part 4: REVIEW Your Organization’s Compliance with FSMA 204
October 12, 2023
The January 20, 2026 FDA Food Traceability Rule, FSMA 204, enforcement date is rapidly approaching. If you’ve been following our Timeline to FSMA 204 Compliance series, you’re familiar with the Learn, Plan, Do, Review strategy we’ve referenced in Parts 1-3 of this series. Part 4 will discuss the timeline’s final but ongoing phase, “Review.”
If you missed Parts 1-3 of our series, you can view them here:
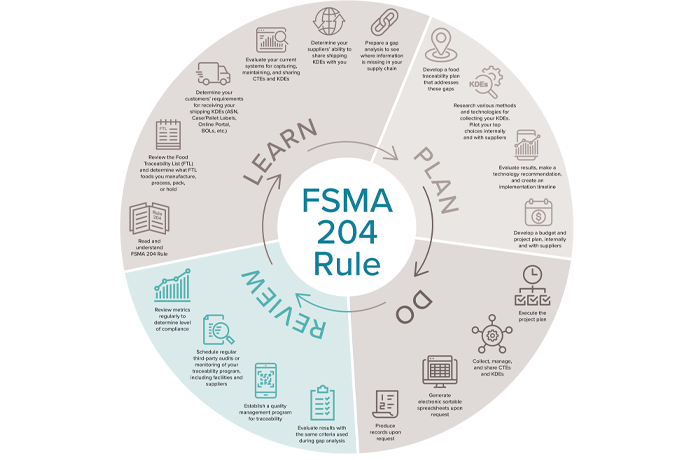
It is a big accomplishment to achieve end-to-end traceability and fully comply with FDA’s Food Traceability Rule. Still, your organization should regularly review and update your processes as needed. We recommend adding these four actions to your traceability plan:
1.Evaluate results with the same criteria used during gap analysis.
Supply Chain Role this applies to: Grower-Packer-Shipper, Processor, Distributor, Grocery Retail & Foodservice
The “Part 1: Learning” phase of this series wrapped up by discussing the importance of preparing a gap analysis. Now that your new processes and systems are in place and have been executed, review those gaps you identified earlier:
- Do your processes still fit customer expectations, or do you need to adjust?
- Do you still have suppliers who aren’t sending their Key Data Element (KDE) records?
- When suppliers send you their KDE records, can you properly ingest their data?
- Do your new systems and processes enable you and your trading partners to fully comply with FSMA 204?
It’s a good idea to revisit these points on a regular basis and ensure compliance requirements (PDF download) are still being met.
2.Establish a quality management program for traceability.
Supply Chain Role this applies to: Grower-Packer-Shipper, Processor, Distributor, Grocery Retail & Foodservice
Establish a monitoring program to measure accuracy, capture errors, and create corrective actions. Leverage internal and third-party auditors to verify the accuracy of your program, then make it part of your ongoing traceability program. Any procedures you employ for data capture, monitoring, and sharing should be reflected in your Traceability Plan. Remember, traceability is a journey, not a destination. Continuous improvement and monitoring are required to assure data quality and confidence in your traceability system.
3.Schedule regular third-party audits or monitoring of your traceability program, including facilities and suppliers.
Supply Chain Role this applies to: Grower-Packer-Shipper, Processor, Distributor, Grocery Retail & Foodservice
Get post-implementation assurance that your systems and processes continue to meet the FDA’s Food Traceability Rule requirements. We recommend impartial, expert support from consultants like New Era Partners, who will provide audit tools and services that may be performed quarterly, semi-annually, or annually, depending on the organization and commodity risk.
4.Review metrics regularly to determine level of compliance.
Supply Chain Role this applies to: Grower-Packer-Shipper, Processor, Distributor, Grocery Retail & Foodservice
Regularly reviewing your traceability metrics will help you recognize gaps or areas where you need to adjust processes. Ensure existing suppliers are still sending their shipping KDEs, and ensure new ones know your organization’s requirements. It’s a good idea to offer regular staff training to ensure your team is consistently and accurately collecting the required data. By reviewing metrics in cohesive dashboards, you’ll more easily notice areas of concern.
Go Beyond Compliance
Now that you are capturing, maintaining, and sharing traceability data, you’ve set the foundation to go beyond compliance and gain ROI from your efforts.
- Reduce food waste: Trace your product to its source to better predict quality and shelf life. Gain visibility into best by, pack, and harvest dates to understand your product beyond the supplier’s data.
- Utilize the compliance team and expertise you have in place to manage traceability for other enterprise-level projects within your organization.
- Achieve a competitive advantage: Connect your supply chain data to gain insights that will give you a competitive advantage with regards to sourcing, quality, food safety, and pricing.
Conclusion
FSMA 204 compliance will never be something you can check off the list, but now that it’s built into your organization’s SOP, maintenance doesn’t have to be a burden. Check in regularly and make small changes as needed along the way.
iFoodDS is the leader in food supply chain traceability solutions. Request a consultation to learn how we can help guide your organization through FSMA 204 compliance.