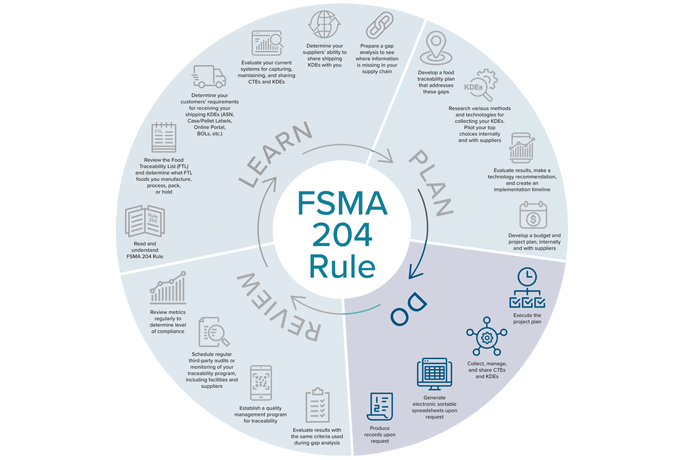
Timeline to FSMA 204 Compliance, Part 3: DO (Put Your FSMA 204 Compliance Plan Into Action)
September 28, 2023
With just over two years until the FDA Food Traceability Rule enforcement date, it’s crucial to prepare early, especially for larger organizations with more complex supplier networks.
You don’t have to be ready to comply overnight, but you should be well on your way to having a plan in place.
Andy Kennedy, Principal Traceability Advisor at New Era Partners and co-writer of FSMA 204, designed a timeline outlining the path to compliance. By now you should’ve learned how the rule applies to your organization, created your traceability plan, and identified your method for capturing, storing, and sharing KDEs. Now it’s time to act and DO all the work you set out to do.
1. Execute the project plan
Supply Chain Role this applies to: Processor, Distributor, Grocery Retail & Foodservice
In the planning phase, you developed a path forward to fill any gaps you discovered from:
- Auditing your supply chain partners
- Evaluating your own systems, and
- Considering required updates to your processes and systems
That path forward should have led you to create your organization’s plan for complying with FSMA 204. In that plan, you identified:
- If your systems can capture, store, and share Key Data Elements (KDEs), and if not, what action would you take to ensure they do so?
- If your trading partners are currently sending, or able to send, all required shipping KDEs, and if not, how would you work with them so that they could do so?
- If you can ingest KDEs from your trading partners, and if not, how might you adjust your processes or improve your systems to do so, or how might you require your trading partners to adjust their processes?
- If your customers have data-sharing requirements that you’re unable to meet, and if so, how would you meet those requirements?
- If you can create an electronic sortable spreadsheet with the FSMA 204-required records within 24 hours, and if not, your plan for doing so.
If the process you already had in place allowed you to do all of the above, you may not have had to make many changes to your organization’s internal processes or those you share with your trading partners.
Implementing your new solution
If not, you’re likely in the implementation phase with your new process or solution provider. This is a critical step in executing your plan. A smooth implementation process, including proper staff training, will prepare your organization for successful execution. You’ll want to ensure:
- Your new solution provider has fully satisfied your organization’s requirements and fulfilled their promises. It is best to address any concerns you may have during implementation and testing.
- Your staff is adequately trained and set up with the necessary devices. Once they start using it, ask for feedback. Make sure they’re comfortable using your new solution.
- Reports, dashboards, and record storage are set up to your standards and the right people have access. If the FDA comes knocking on your door, would someone be able to pull that electronic sortable spreadsheet within 24 hours?
- Your new processes and training procedures are documented so that you can quickly train new staff as they are hired.
Remember that FSMA 204 compliance relies on everyone doing their part. This is the time to make sure everyone understands their role.
Compliance doesn’t happen overnight. Like our compliance timeline conveys, collecting, managing, and sharing KDEs is not a small task and should be approached in steps. Think of each as a step towards full supply chain visibility.
2. Collect, manage, and share CTEs and KDEs
Supply Chain Role this applies to: Processor, Distributor, Grocery Retail & Foodservice
Processes are in place, and the staff is trained. Now it’s time to consistently collect KDEs from suppliers, store and manage those KDEs in your internal systems, and share them with your customers. If you need a refresher on which Critical Tracking Events (CTEs) you are responsible for and which KDEs are required for each CTE, visit FDA.gov (view PDF).
We recommend a step-by-step approach:
- Start capturing your suppliers’ shipping KDEs. By now, you should have identified any suppliers who could not share their KDEs with you and found a solution for them to do so. If some are still non-compliant, it’s time to work with them to ensure they are or consider ending the relationship.
- Start recording your own receiving KDEs. You have the processes and tools in place now to do this. Your suppliers are sending their shipping KDEs, which means you already have most of the KDEs you need to record your receiving KDEs.
- Understand where your shipping data is stored in your internal systems so that you can pull an electronic sortable spreadsheet within 24 hours when asked by FDA. This data can be useful in other ways, too. Start thinking about how you might use it to make proactive decisions.
- Start capturing your shipping KDEs and ensure you can send them to your trading partners, if applicable. Ensure your method of sending shipping KDEs meets their requirements if they’re an external partner not using the same system as your organization.
- If applicable, start collecting and storing transformation KDEs.
- Write your Traceability Plan. FDA requires this for any organization that is covered by FSMA 204. View the list of requirements and our example of a Traceability Plan.
3. Generate electronic sortable spreadsheets upon request
Supply Chain Role this applies to: Grower-Packer-Shipper, Processor, Distributor, Grocery Retail & Foodservice
We recommend going through the process of creating your electronic sortable spreadsheet now, even if one hasn’t been requested by FDA. Make sure you have identified more than one person who can do this, in case your main point of contact is on vacation, leaves the company, or is otherwise unavailable.
4. Produce records upon request
Supply Chain Role this applies to: Grower-Packer-Shipper, Processor, Distributor, Grocery Retail & Foodservice
It’s a good idea to double-check that you have all necessary records that may be needed supporting the information in the electronic sortable spreadsheet. FDA may ask for additional details after the 24-hour period, so it’s a good idea to know where to find the information.
Conclusion:
You’ve now achieved end-to-end traceability and should feel confident that your organization is FSMA 204 compliant. You have the foundation to go beyond required data capture, turning regulatory compliance into a profit opportunity with a complete understanding of end-to-end fresh food. Request a consultation with iFoodDS to learn how to take your organization to the next level.
Read our complete Timeline to FSMA 204 Compliance article series:
- Part 1: Learn
- Part 2: Plan
- Part 3: Do (read this article)
- Part 4: Review